Ketamine and Cocaine: Risks of Mixing Cocaine & Ketamine
Mixing cocaine and ketamine, sometimes called “Calvin Klein” or “CK” in nightlife settings, is dangerous because it combines a potent stimulant with a dissociative anesthetic in a way that impairs self-monitoring, obscures overdose warning signs, and amplifies cardiovascular, psychiatric, and respiratory risks.
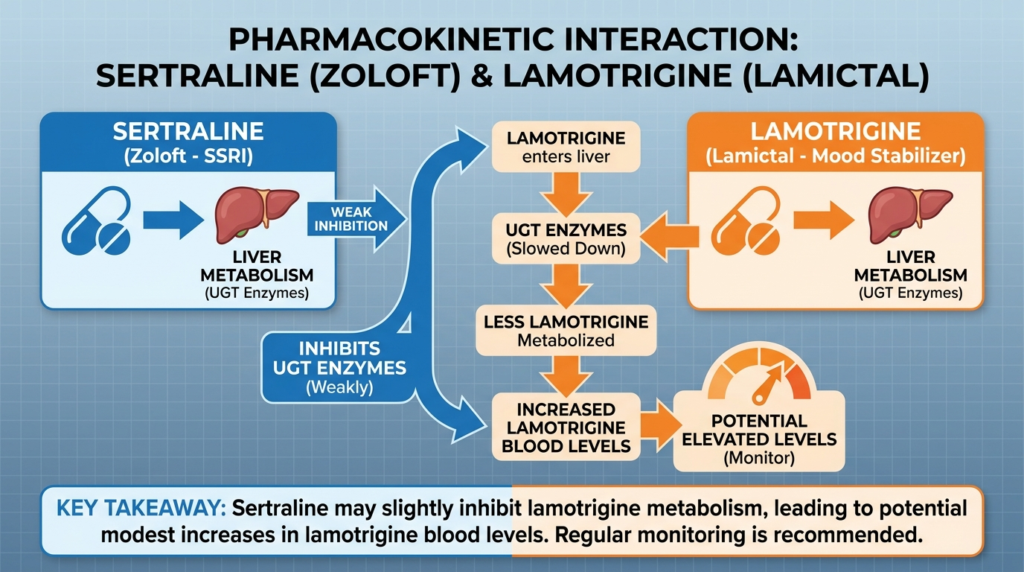
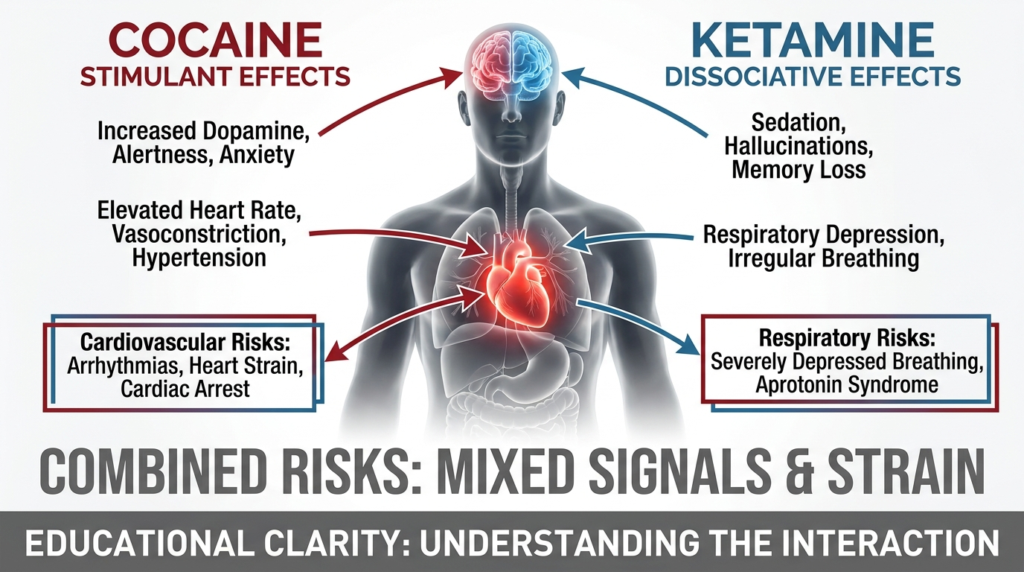
Cocaine drives heart rate, blood pressure, and agitation while ketamine distorts perception and consciousness, creating an unstable state where users may feel less impaired than they actually are. Emergency data show that cocaine-positive patients given ketamine for severe agitation had nearly six times the intubation rate of those without cocaine, signaling a serious interaction risk.
This article explains why the combination is especially hazardous, what happens in the body when these drugs are mixed, and what to do if someone shows signs of overdose.
Why People Mix Cocaine and Ketamine?
The combination of cocaine and ketamine has gained visibility in club, festival, and electronic dance music scenes, where it is often sought for a perceived “balance” between stimulation and detachment. Users report mixing the two to intensify euphoria, stay alert while softening cocaine’s harsh edge, or create a feeling of being energized yet emotionally grounded.
The slang term “Calvin Klein” derives from the initials C and K rather than any pharmacological concept, and the nickname itself can make the practice seem stylized or controlled when it is actually medically unstable.
Nightlife environments reward certain states, sociability, endurance, altered sensory experience, and wakefulness over many hours, and cocaine and ketamine may seem functionally complementary in those settings.
Cocaine supports energy and confidence; ketamine adds surreal detachment and altered perception. Yet those same settings also increase risk through overheating, dehydration, repeated dosing, alcohol co-use, and delayed emergency response in loud, chaotic environments.
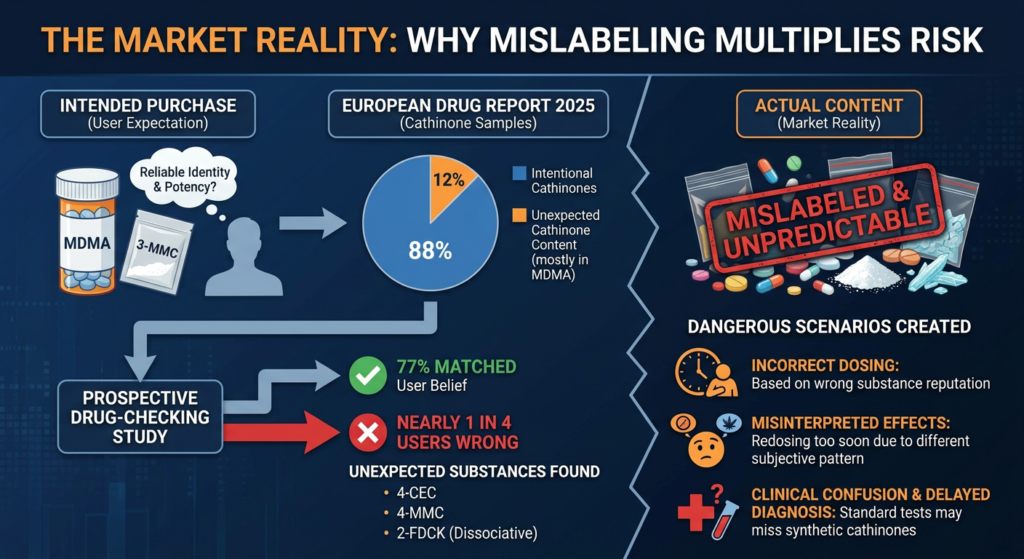
Recent reporting drawing on European drug trends suggests that ketamine availability has risen in parts of Europe while cocaine purity has increased significantly over the last decade.
These market shifts make the combination both more accessible and more dangerous, as higher purity reduces the margin for dosing error, especially when users are already mixing substances intentionally.
What Happens When You Mix Cocaine and Ketamine?
When cocaine and ketamine are taken together, the body does not simply average the two effects into a safer middle state.
Instead, the combination produces simultaneous cardiovascular stimulation and perceptual disruption, impaired awareness of toxic warning signs, and reduced ability to self-monitor.
Users may feel less impaired than they are or less physically threatened than they are, which can promote redosing and delay help-seeking.
Cocaine’s Effects on the Body
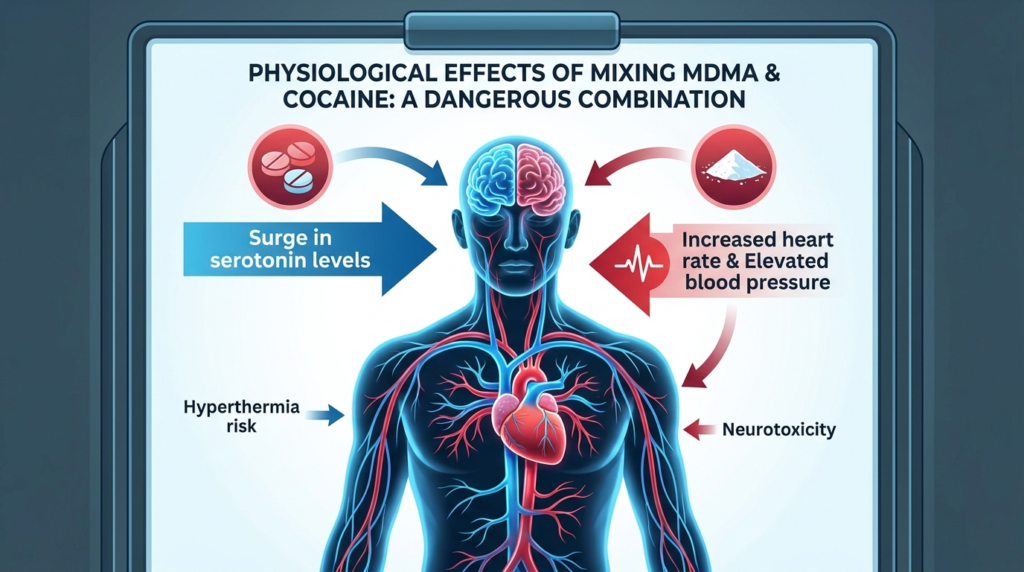
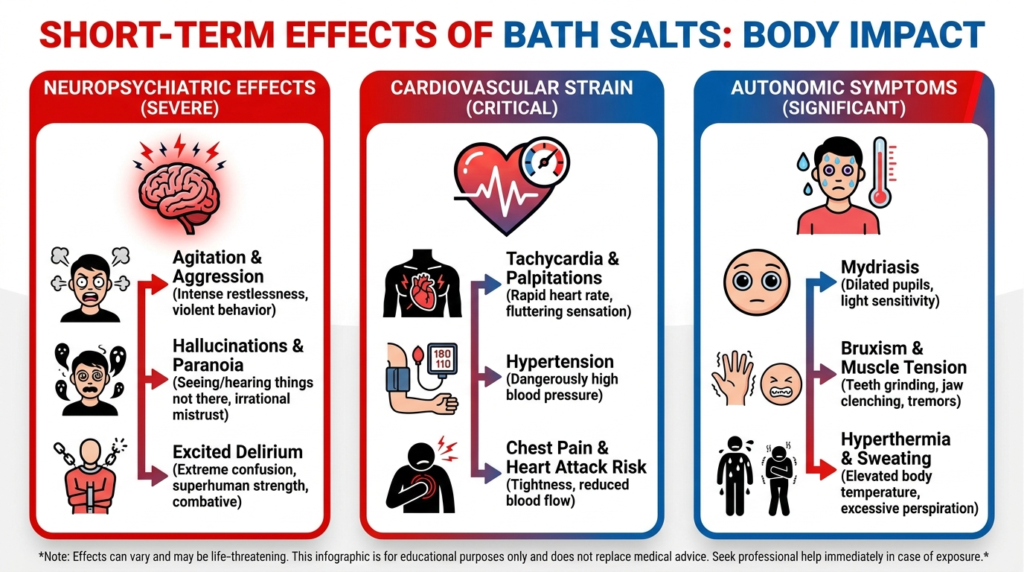
Cocaine is a potent stimulant that increases catecholaminergic activity and produces a classic sympathomimetic toxidrome. Acute intoxication can include tachycardia, hypertension, diaphoresis, hyperthermia, agitation, seizures, tachyarrhythmias, myocardial ischemia, stroke, and psychotic symptoms.
Cocaine also has sodium-channel-blocking effects, which can produce local anesthetic properties, QRS widening, and seizure risk, distinguishing it from a simple stimulant and adding arrhythmic danger in overdose.
Fever in cocaine intoxication may result from both peripheral vasoconstriction and central dysregulation of temperature. Hyperthermia is especially dangerous because it can trigger organ injury, rhabdomyolysis, coagulopathy, seizures, and collapse.
In festivals or clubs where dancing, crowding, dehydration, and prolonged wakefulness are common, this risk may increase further.
Ketamine’s Effects on the Body
Ketamine is a dissociative anesthetic used medically but also misused recreationally. Acute toxicity can involve dissociation, confusion, hallucinations, psychosis, impaired consciousness, shallow or distressed breathing in overdose, and airway risk requiring supportive management.
There is no recognized antidote for ketamine overdose. Emergency treatment is primarily supportive, centered on airway, breathing, and circulation, with possible use of activated charcoal in selected circumstances, symptom-directed medications such as benzodiazepines, alpha-2 agonists, or anticholinergics, IV fluids, and monitoring even after symptoms appear to improve.
Ketamine can also be difficult to recognize in mixed intoxication because symptoms overlap with other substances and routine toxicology may omit ketamine unless specifically screened for.
The Dangerous Interaction
The central problem is not that cocaine and ketamine produce opposite effects in a neat, canceling way. The evidence instead suggests they can produce stacked instability. Cocaine increases autonomic stress while ketamine alters perception, coordination, and insight.
Each can independently contribute to agitation, hallucinations, and psychosis under some circumstances. This means the user may move quickly between stimulation, dissociation, panic, agitation, confusion, and collapse. Such a presentation is harder for bystanders to interpret and more difficult for clinicians to categorize than a straightforward single-drug overdose.
One of the most clinically striking pieces of evidence comes from a prehospital study of patients who received intramuscular ketamine for excited delirium. In that cohort, suspected cocaine intoxication was associated with a significantly higher risk of subsequent emergency department intubation, with an odds ratio of 5.75.
Among 27 patients with suspected cocaine intoxication, nine were intubated, compared with four of 50 without cocaine intoxication. This study does not prove that all recreational cocaine plus ketamine use always causes intubation, but it provides a meaningful safety signal that when cocaine and ketamine co-occur in already unstable patients, airway deterioration requiring intubation becomes materially more likely.
Acute Risks and Side Effects of Mixing Cocaine and Ketamine
The combination is especially dangerous because several hazards converge. A common misconception is that a stimulant can cancel out a sedative or dissociative. In practice, one drug may mask warning signs of the other without preventing toxicity.
Cocaine may make someone feel more awake, but it does not protect against ketamine-induced disorientation, psychosis, poor coordination, or overdose. Ketamine may blunt some of cocaine’s harsh subjective edge, but it does not reduce cocaine’s cardiovascular or thermoregulatory toxicity.
Cardiovascular and Thermoregulatory Dangers
Cocaine raises heart rate, blood pressure, and arrhythmia risk. Acute cocaine intoxication can cause hypertension, tachycardia and tachyarrhythmias, coronary vasospasm, myocardial ischemia or infarction, and stroke and intracranial hemorrhage. These effects can occur even in younger individuals without traditional cardiovascular risk factors.
The user may therefore experience severe autonomic strain while being too dissociated or confused to recognize chest pain, palpitations, overheating, or impending collapse. This pairing of physiologic stress with impaired insight is one reason the combination is more dangerous than either drug in a fully self-aware user.
Psychiatric and Behavioral Toxicity
Recent overdose research found that agitation, aggression, anxiety, hallucinations, and psychosis were frequent in overdose presentations and were significantly associated with cocaine and other stimulants. Psychosis was specifically associated with cocaine detection. This is highly relevant because users may mix cocaine with ketamine seeking calm or grounding, yet cocaine is one of the substances most strongly tied to behavioral toxicity in overdose populations.
Ketamine can produce dreamlike detachment, hallucinations, and altered perception. In low-to-moderate recreational use, this may be subjectively sought after. In higher or mixed doses, it can become disorienting, frightening, and unsafe.
The person may misinterpret surroundings, lose balance, panic, or become unable to communicate accurately. The combination can produce a dangerous psychiatric profile because cocaine can intensify paranoia, urgency, and overactivation while ketamine can distort reality-testing and bodily awareness. Together they can increase the chance of panic, aggression, confusion, or bizarre behavior.
Excited delirium is not merely acting erratically. It is a medical emergency associated with hyperthermia, rhabdomyolysis, collapse, respiratory arrest, and cardiorespiratory arrest. Cocaine-related hyperthermic syndromes can include hallucinations, agitation, and excited delirium with progression to collapse and arrest. The relevance to cocaine-ketamine mixing is direct: ketamine may intensify altered sensorium while cocaine drives the adrenergic crisis.
Respiratory and Airway Compromise
Public-facing overdose guidance on ketamine highlights shallow breathing, respiratory distress, and unconsciousness as warning signs requiring emergency care. Although cocaine is stimulating, severe intoxication can progress to collapse, irregular breathing, seizures, and cardiorespiratory arrest. In hyperthermic excited delirium, respiratory arrest can be part of terminal deterioration.
The strongest direct concern comes from the prehospital study whose findings suggest cocaine-positive status may increase intubation risk after ketamine in severe agitation. Combined with the known airway focus of ketamine overdose management, this supports a cautious conclusion: airway and breathing problems should be anticipated early in severe cocaine-ketamine intoxication, even if the patient initially appears amped up rather than sedated.
Impaired Overdose Recognition
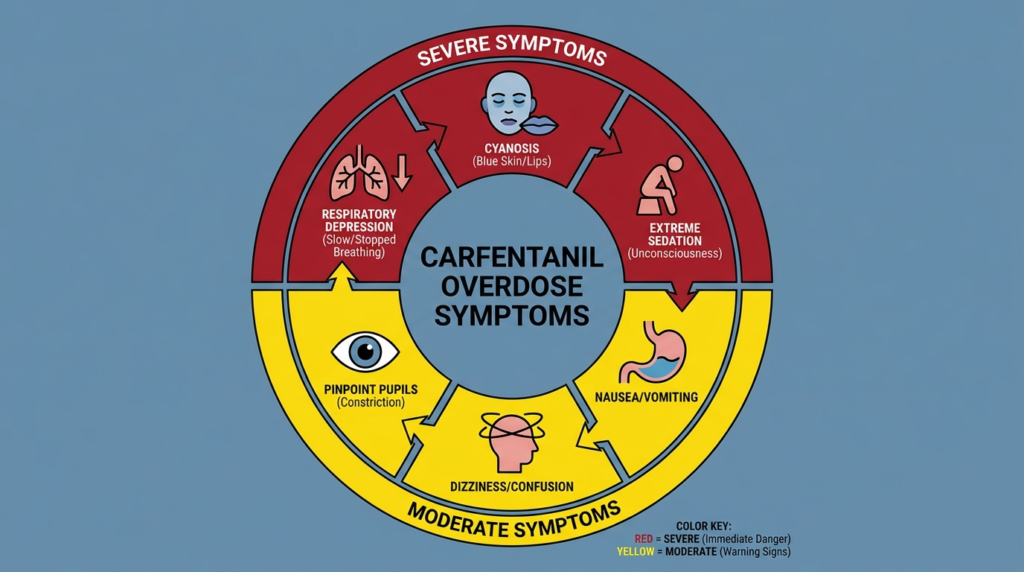
Bystanders often expect overdose to look like opioid overdose: stopped breathing, blue lips, pinpoint pupils, unresponsiveness. Cocaine-ketamine overdose may look very different, severe confusion, hallucinations, erratic or aggressive behavior, chest pain, rapid pulse, extreme overheating, shallow breathing later in the course, intermittent responsiveness, or collapse after prolonged agitation. This variability can cause dangerous delays.
People in overdose frequently underreport co-ingested substances. If the user says they only took ketamine, cocaine may be missed; if they say they only did cocaine, ketamine may be missed. Witnesses may also assume one substance based on reputation or paraphernalia rather than actual exposure.
Users seeking a balanced high may interpret warning signs as expected effects: derealization interpreted as good ketamine, racing heart interpreted as just cocaine, confusion interpreted as temporary, agitation seen as part of the party environment. This misattribution is a major danger.
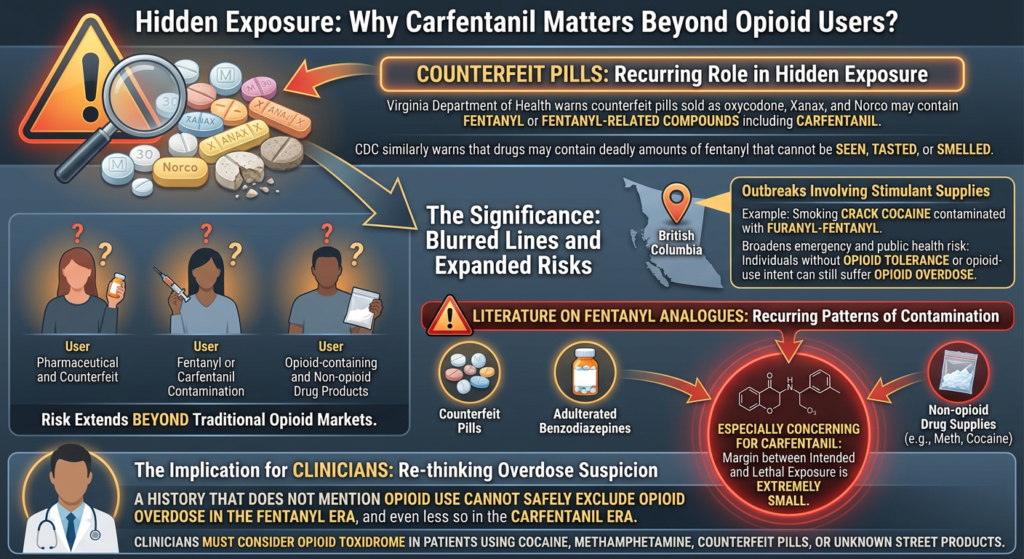
Polysubstance Use and Hidden Contamination Risks
The strongest contextual evidence comes from an Irish survey of 1,193 adult festival attendees. In that cohort, 96 percent reported drug use, 86.8 percent reported polysubstance use, and participants reported using an average of three substances at one time. Common combinations involved alcohol, cocaine, ketamine, and MDMA.
This is crucial because cocaine-ketamine use does not usually occur in isolation. In nightlife settings, people often combine multiple substances to engineer a certain experience, cope with comedowns, prolong wakefulness, or modulate unwanted effects.
Multiple sources indicate that ketamine misuse often occurs with other substances, including alcohol, cocaine, and MDMA. One review specifically notes that in acute ketamine toxicity, simultaneous use of alcohol, cocaine, and MDMA has been reported. Another recent review argues that recreational ketamine use is frequently combined with other substances and that routine toxicology may miss ketamine in mixed intoxications if it is not specifically screened for.
In acute recreational drug overdose, self-report is often inaccurate, with major underreporting documented for benzodiazepines, pregabalin, and THC.
This finding implies a broader lesson: clinicians and bystanders should assume that some co-ingestants may be undisclosed, forgotten, unknown, or denied. In suspected cocaine-ketamine intoxication, this means the true exposure may include additional depressants or stimulants, substantially altering risk.
Polysubstance use increases danger through several pathways: additive or synergistic toxicity, masking of warning signs, impaired ability to judge dose or deterioration, more difficult emergency management, contaminated or mis-sold substances, and delayed recognition by bystanders and clinicians.
For cocaine-ketamine specifically, the problem is intensified by the fact that one drug can make the user feel more stimulated while the other impairs perception and judgment.
Warning Signs of Cocaine and Ketamine Overdose
Seek immediate emergency care for difficulty breathing or shallow breathing, chest pain, severe confusion, psychosis or extreme agitation, seizures, hyperthermia or overheating, unresponsiveness, blue or gray discoloration, or irregular pulse. A cocaine overdose may look like severe agitation, chest pain, overheating, tachycardia, or seizure.
A ketamine overdose may look like confusion, hallucinations, shallow breathing, unresponsiveness, or dissociation. A mixed overdose may show features of both or shift between them. This can confuse bystanders into assuming the person is just really high, especially if they are intermittently responsive or moving around.
The combination is especially dangerous because it can look less immediately recognizable than classic opioid overdose while still becoming life-threatening.
What to Do in an Emergency?
If ketamine overdose is suspected, bystanders are advised to call 911 immediately, assess responsiveness, breathing rate, and pulse, monitor the person while awaiting emergency medical services, and provide medical personnel with details about suspected substances, symptoms, and relevant history. This advice is applicable to suspected cocaine-ketamine overdose as well.
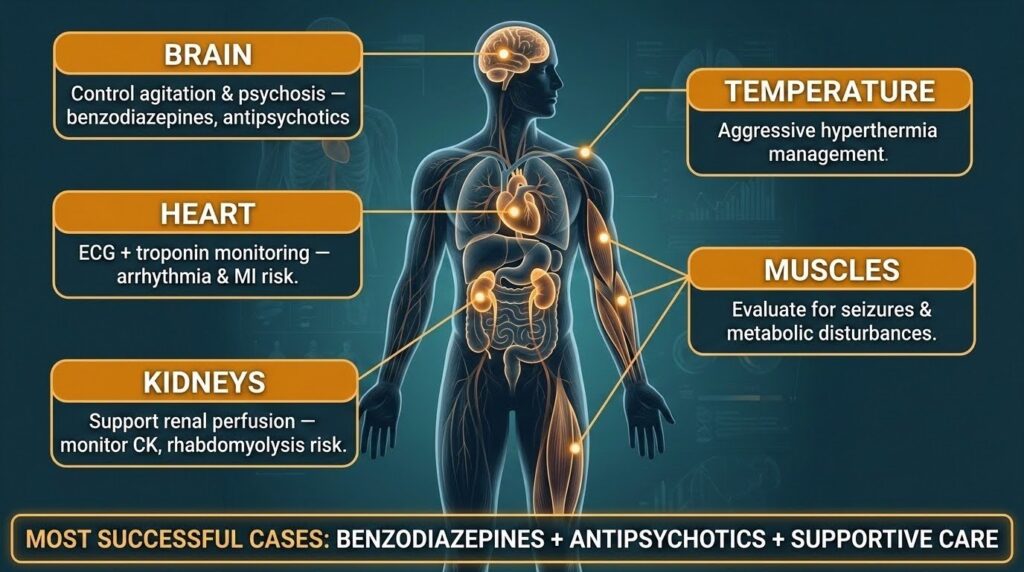
Standard initial treatment for acute cocaine toxicity includes benzodiazepines, active cooling, and attention to end-organ damage, trauma, and seizures. Many cocaine toxicity manifestations, especially agitation, tachycardia, and hypertension, improve with sedatives. IV benzodiazepines are first-line; antipsychotics may be added if agitation remains uncontrolled.
Because there is no antidote for ketamine, overdose management centers on airway, breathing, circulation, supportive care, IV crystalloids for dehydration or hyperthermia, observation and monitoring after symptom improvement, and symptom control with medications as needed.
The combination may require simultaneous management of violent agitation or psychosis, hyperthermia, hypertension and tachycardia, respiratory compromise, possible seizures, and trauma from falls, restraint, or combative behavior. This is one reason the combination is especially dangerous: it can force clinicians to treat both a stimulant emergency and a dissociative overdose picture at once.
Naloxone does not reverse cocaine or ketamine directly. However, because stimulant drugs can be contaminated with opioids such as fentanyl, public health sources recommend considering naloxone when opioid involvement is suspected. This is relevant in uncertain mixed overdoses where breathing is impaired.

Harm Reduction and Risk Reduction
The surest risk reduction is not mixing cocaine and ketamine at all. Based on the evidence, this is the clearest recommendation. Because co-ingestion is common and self-report is unreliable, do not assume only one drug was taken, monitor for both stimulant and dissociative effects, and expect hidden substances or contamination.
Given the strong role of setting, avoid using alone, avoid repeated redosing, avoid combining with alcohol, benzodiazepines, or unknown powders, cool down, hydrate, and rest, and use drug-checking where available. Festival research indicates that drug-checking services can prompt safer behavior such as warning others, being more careful with polysubstance use, and taking lower doses.
Routine toxicology may miss ketamine, and users often do not know what is actually in a substance. Drug checking is not a guarantee of safety, but it can reduce some uncertainty and may support harm-reduction decisions in nightlife settings.
Long-Term Health Concerns
Although this report focuses on acute risk, some chronic implications are relevant. Ketamine misuse with multiple substances is associated with higher overdose and fatality risk. Cocaine use carries cumulative cardiovascular and psychiatric harms. Repeatedly combining the two may therefore increase both acute-event risk and longer-term substance use disorder burden.
Frequent ketamine use has been associated with urinary tract and bladder complications, cognitive problems, sleep issues, and depression-related concerns. Chronic stimulant use can contribute to recurrent emergency visits, mental health deterioration, cardiovascular complications, and ongoing relapse cycles that strain both individuals and health systems.
Why This Combination Deserves Serious Attention?
Mixing cocaine and ketamine is especially dangerous because it combines the cardiovascular, thermoregulatory, neurologic, and psychiatric hazards of cocaine with the dissociative, perceptual, and airway-related hazards of ketamine in a way that impairs self-monitoring, obscures overdose recognition, and complicates emergency management. This is not merely a generic warning about any drugs mixed together. It is a specific high-risk pairing.
Cocaine alone is medically dangerous enough to cause myocardial infarction, stroke, seizures, hyperthermia, excited delirium, and psychosis. Ketamine alone can impair consciousness, perception, and breathing, and has no antidote; treatment is supportive and airway-focused. The combination is often used intentionally to balance effects, but that subjective balance is misleading because one drug can mask the warning signs of the other rather than neutralize toxicity.
In real-world settings, cocaine and ketamine are rarely isolated exposures. Polysubstance use is the norm in festival and nightlife environments, and overdose self-report is often inaccurate. There is direct human clinical evidence of higher intubation risk in cocaine-positive patients exposed to ketamine in severe agitation settings. While not definitive for all recreational scenarios, this is a serious warning signal.
For these reasons, the combination deserves to be viewed not as a trendy club drug practice but as a medically unstable form of polysubstance exposure with a plausible path to psychosis, hyperthermia, collapse, airway compromise, and death.
If you or someone you care about is struggling with cocaine, ketamine, or polysubstance use, our professional help can make all the difference. Thoroughbred Wellness & Recovery offers compassionate, evidence-based care for substance use and co-occurring mental health conditions in a supportive environment.
Our team is available 24/7 to answer your questions and guide you toward lasting recovery. Contact our team today to learn how we can help.